Abecma (idecabtagene vicleucel) is a prescription therapy used in specific cases of multiple myeloma. This medication is prepared as a suspension (liquid mixture) created from your own immune cells. It is administered as an intravenous (IV) infusion into a vein under medical supervision.
ABECMA CAR T therapy is indicated for adults with multiple myeloma that:
- has relapsed after previously responding to treatment, or
- has not responded adequately to prior therapy (or has stopped responding)
Before your doctor can prescribe ABECMA CAR T, you must have already received at least two different treatments for multiple myeloma.
For additional details about how this therapy works, see the “What is Abecma used for?” section below.
Abecma basics
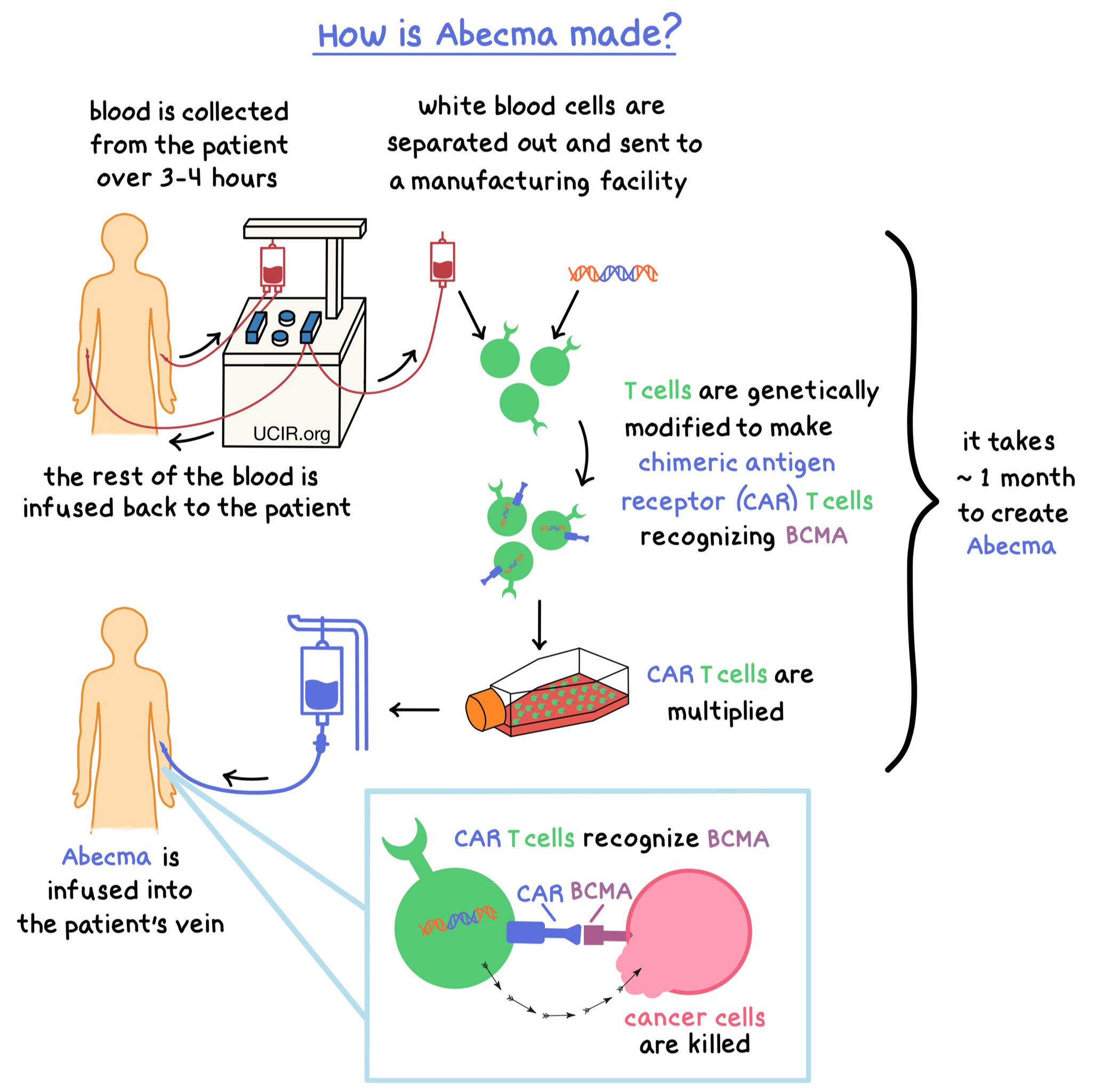
Abecma contains the active ingredient idecabtagene vicleucel. It belongs to a class of medications known as CAR T-cell therapies, a specialized form of immunotherapy designed to harness your immune system to fight cancer. These treatments are manufactured using your own white blood cells.
Abecma is a biologic drug. Biologic medications are produced from components of living organisms. Abecma is available only as a brand-name product and does not have a biosimilar equivalent.
Biosimilars are comparable to generic drugs, but while generics are made for nonbiologic medications, biosimilars are developed for biologic therapies.
Abecma carries boxed warnings, which are the most serious warnings issued by the Food and Drug Administration (FDA). These warnings are discussed in detail in the “What should be considered before receiving Abecma?” section.
What is Abecma used for?
ABECMA CAR T is approved for certain adults with multiple myeloma. Specifically, it is used when the disease:
- has returned after prior successful treatment, or
- has not improved with previous therapy (or has stopped responding)
If you have undergone at least two prior treatment regimens for multiple myeloma, your doctor may consider Abecma. These earlier treatments may have included:
- an immunomodulatory agent
- a proteasome inhibitor
- an anti-CD38 monoclonal antibody
Multiple myeloma is a cancer that develops in plasma cells within the bone marrow. In early stages, symptoms may be minimal. As the disease advances, it can lead to complications such as anemia, kidney dysfunction, or bone damage due to uncontrolled growth of abnormal plasma cells.
ABECMA CAR T therapy is created from T cells collected from your bloodstream. (T cells are a type of white blood cell.) In a laboratory, these cells are genetically modified to recognize and attach to the B-cell maturation antigen (BCMA) found on myeloma cells. You can learn more about the Abecma target antigen and how BCMA supports cancer cell survival and growth. When the engineered T cells bind to BCMA, they destroy the cancerous cells.
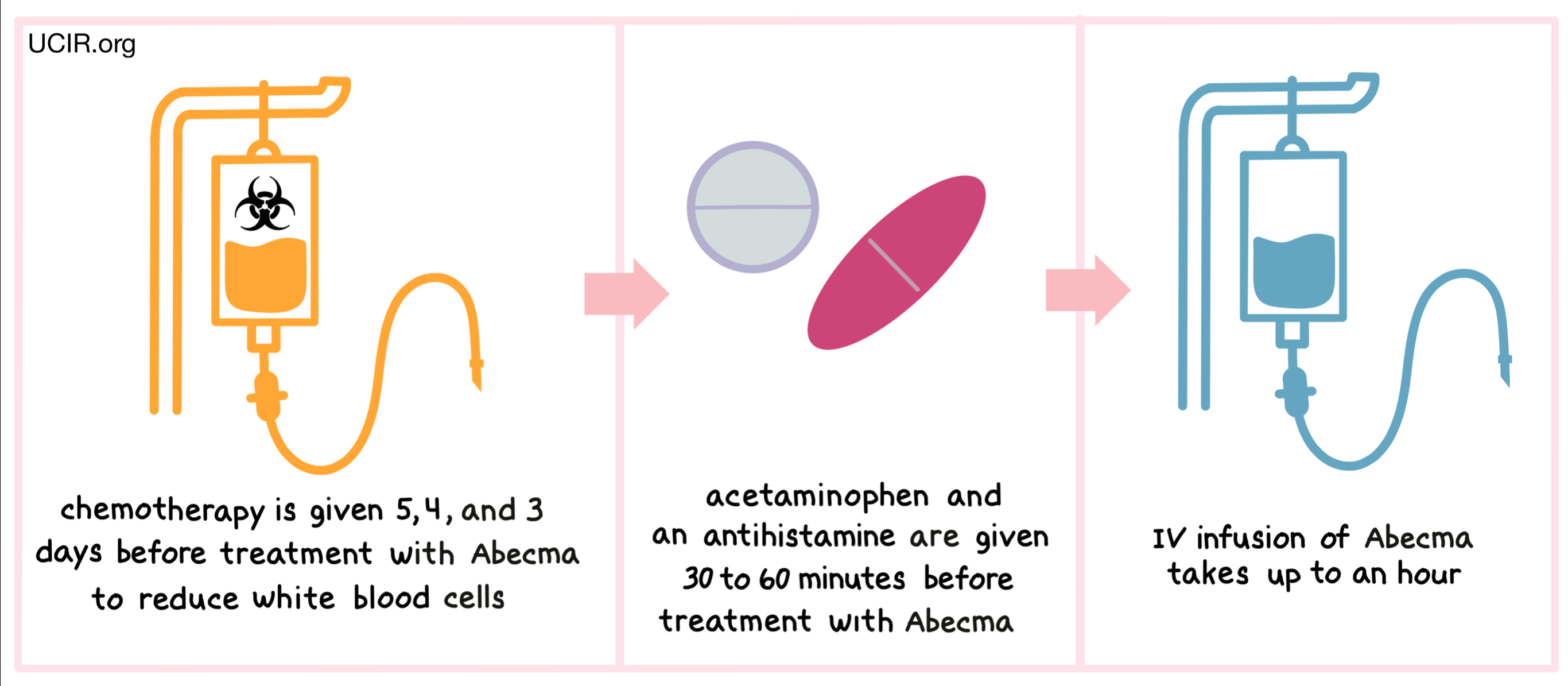
Although Abecma is delivered as a one-time infusion, additional medications are required as part of the treatment protocol:
- Chemotherapy drugs: Cyclophosphamide and fludarabine are given for 3 consecutive days before Abecma. The infusion of Abecma typically takes place 2 to 7 days after completing chemotherapy.
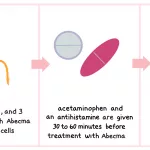
- Acetaminophen (Tylenol) and diphenhydramine: These medications are administered 30 to 60 minutes prior to the infusion to help reduce the likelihood of infusion-related side effects.
What are some frequently asked questions about Abecma?
Below are answers to common questions about ABECMA CAR T therapy.
How does Abecma compare with Carvykti?
Both Abecma and Carvykti (ciltacabtagene autoleucel) are prescribed for multiple myeloma that:
- has relapsed after earlier effective treatment, or
- has not responded to prior therapy (or has stopped responding)
Abecma is generally considered after at least two previous treatment regimens. In contrast, Carvykti may be prescribed after at least one prior therapy.
Each medication is prepared as a suspension and administered as a single intravenous (IV) infusion. You receive only one infusion of either therapy.
Both are CAR T-cell therapies derived from your own immune cells, but they contain different active ingredients. Abecma contains idecabtagene vicleucel, whereas Carvykti contains ciltacabtagene autoleucel.
Your healthcare provider can help determine which option may be most appropriate for your situation.
Can Abecma be used for other types of blood cancer?
Abecma is approved solely for multiple myeloma that:
- has relapsed after previous treatment, or
- has not improved with prior therapy (or has stopped responding)
It is not approved for other blood cancers.
However, its prescribing information includes a boxed warning regarding the potential risk of developing certain other cancers involving T cells. If you undergo ABECMA CAR T therapy, your doctor will continue monitoring you long term for signs of these conditions.
For information about therapies for other blood cancers, consult your doctor.
Abecma cost and savings
The cost of ABECMA CAR T therapy can vary depending on your insurance coverage, treatment plan, and the facility where you receive the infusion.
- Cost information and savings coupons: Visit Optum Perks to view price estimates using available coupons. (Note: Optum Perks coupons cannot be combined with insurance copays or benefits.)
- Savings program: Speak with your doctor about financial assistance. The Cell Therapy 360 program may provide support.
You may also explore additional strategies for reducing prescription costs.
Optum Perks is owned by RVO Health. By clicking on this link, we may receive a commission. Learn more.
What are Abecma’s side effects?
Like many medications, ABECMA CAR T therapy can cause both mild and serious side effects. Not everyone experiences the same reactions.
Side effects may depend on factors such as:
- your age
- other medical conditions you have
- other medications you are taking
Your healthcare provider can explain potential risks and how to manage them.
Note: After FDA approval, side effects continue to be monitored. If you experience a side effect and wish to report it, visit MedWatch or call 800-FDA-1088.
Mild side effects
Some reported mild side effects include:
- fever (with or without low white blood cell counts)
- bone, muscle, or joint pain
- fatigue
- dizziness, tremor, or chills
- headache
- gastrointestinal issues such as nausea, vomiting, diarrhea, or reduced appetite
- swelling in certain areas
- upper respiratory infection
Many mild symptoms improve within days to weeks. Contact your doctor if symptoms persist or worsen.
Serious side effects
Serious side effects have been reported. Seek immediate medical care if you believe you are experiencing a medical emergency.
- hypogammaglobulinemia
- breathing difficulties or low oxygen levels
- low phosphate or sodium levels
- elevated liver enzymes or triglycerides
- blood clotting abnormalities
- kidney problems
- mental status changes such as confusion
- heart rhythm or blood pressure changes
- serious infections (including pneumonia)
- boxed warnings including cytokine release syndrome and neurologic toxicity
What is Abecma’s dosage?
Your doctor will explain your individualized treatment process.
Form and strength
Abecma is produced from your own T cells collected at a specialized center. The cells are sent to a laboratory for modification, a process that usually takes about 4 weeks.
The medication is supplied as a suspension and administered as a single IV infusion.
Recommended dosage
The standard dosage for multiple myeloma is one IV infusion given over at least 30 minutes.
How to receive
You will receive preparatory chemotherapy, followed by your infusion 2 to 7 days later. Acetaminophen and diphenhydramine are given beforehand to reduce side effect risk.
After infusion, you will remain at the treatment center for at least 7 days for monitoring and stay nearby for an additional 2 weeks.
What should be considered before receiving Abecma?
Interactions
No specific drug interactions are listed in the prescribing information, but always inform your doctor about all medications and supplements you take.
Pregnancy and breastfeeding
It is unknown whether Abecma is safe during pregnancy or breastfeeding. Because of its mechanism of action, it may harm a fetus. Pregnancy testing is typically required beforehand.
Boxed warnings
ABECMA CAR T carries boxed warnings for:
- Cytokine release syndrome (CRS) and neurologic toxicity
- Serious immune system conditions
- Prolonged low blood cell counts
- Development of certain other cancers
Due to these risks, close monitoring is required after infusion.
Similar drugs
Other therapies are available for multiple myeloma. Carvykti (ciltacabtagene autoleucel) is another CAR T-cell therapy option. Discuss alternatives with your healthcare provider to determine the most suitable approach.
Disclaimer: HealthEH has made every effort to ensure that all information is accurate, complete, and current. However, this article should not replace professional medical advice. Always consult a qualified healthcare professional before starting any medication. Drug information may change and does not cover all possible uses, precautions, warnings, interactions, or adverse effects.
















Leave a Reply
You must be logged in to post a comment.