CAR T-cell therapy is an advanced form of immunotherapy used to treat multiple myeloma when standard therapies are no longer effective. It has contributed to longer remission periods and improved survival outcomes, although comprehensive long-term safety and durability data are still developing.
Chimeric antigen receptor (CAR) T-cell therapy is a cell-based gene therapy that modifies a patient’s own immune cells to better recognize and attack cancer. Because it enhances the body’s immune response against malignant cells, it is classified as immunotherapy.

CAR T-cell therapy is currently used in certain blood cancers, including multiple myeloma. As interest grows in innovative treatments, many patients also seek information about the Abecma approval date and the broader ABECMA FDA approval history to better understand how these therapies became available.
This article explains how CAR T-cell therapy works for multiple myeloma, what the treatment process involves, possible side effects, and what current research shows about effectiveness.
What is CAR T-cell therapy?
T cells are a type of white blood cell that originate from stem cells in the bone marrow. As a central component of the immune system, T cells identify and eliminate abnormal cells and pathogens. In multiple myeloma, however, T cells often fail to recognize cancer cells as dangerous.
CAR T-cell therapy is designed to correct this immune system blind spot.
With autologous CAR T-cell therapy, your own T cells are used for treatment. The process begins with an intravenous (IV) blood draw. Your blood is circulated through a machine that separates out T cells and then returns the remaining blood to your body. This procedure, known as leukapheresis, typically takes a few hours.
In a specialized laboratory, the collected T cells are genetically engineered to add chimeric antigen receptors (CARs) to their surface. These CARs are proteins that enable T cells to specifically identify targeted tumor cells. After modification, the cells are multiplied in the lab to create sufficient numbers for treatment, a process that may take several weeks. Occasionally, the collection process must be repeated.
Once enough CAR T-cells are produced, they are frozen and transported to the treatment facility. Before infusion, they are thawed and then administered back into your bloodstream. These reprogrammed cells can now detect and attack cancer cells and continue multiplying within the body.
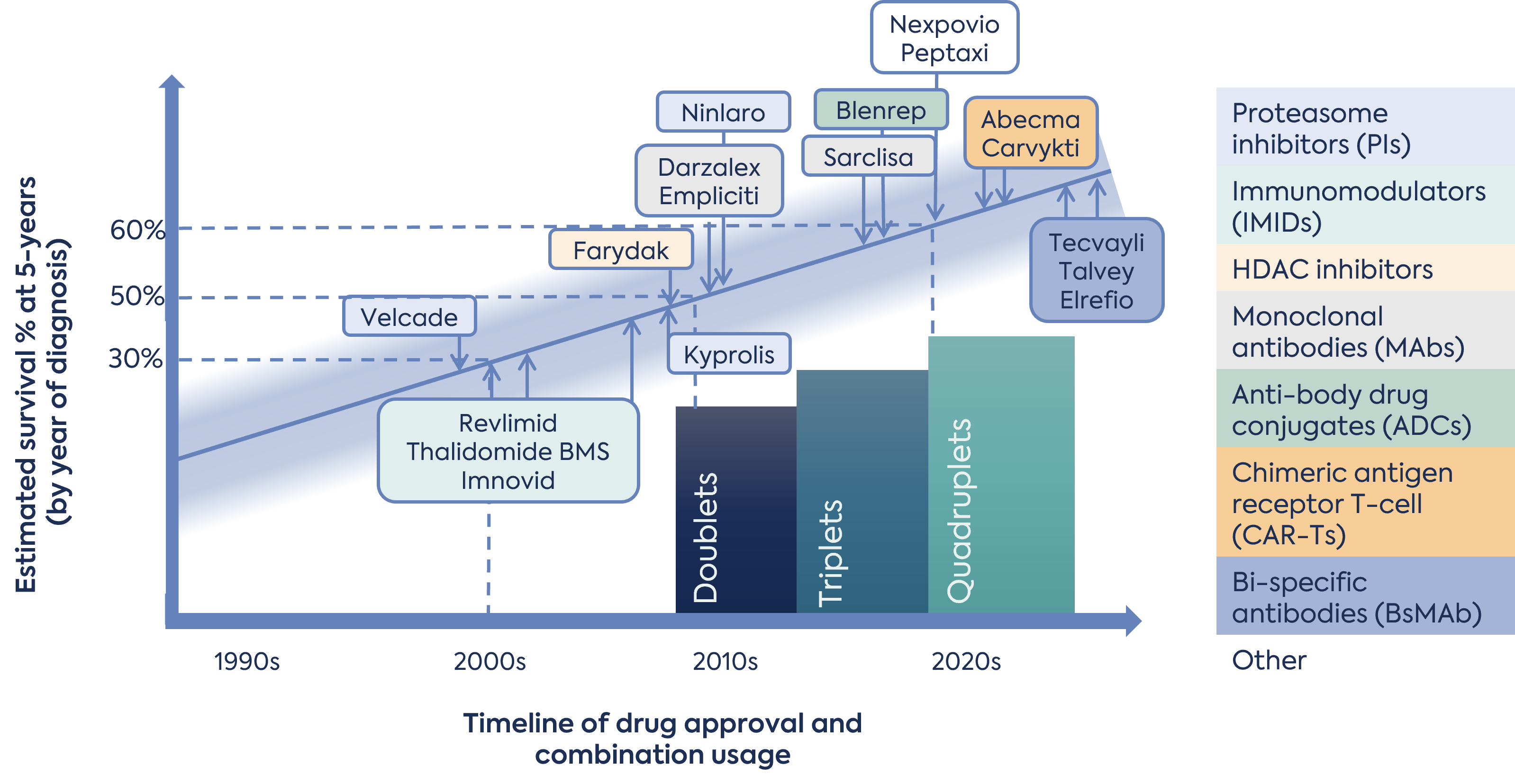
The two CAR T-cell therapies approved for multiple myeloma are idecabtagene vicleucel (Abecma) and ciltacabtagene autoleucel (Carvytki). Patients and clinicians often compare Abecma vs Carvykti when evaluating treatment options, especially in the context of relapsed or refractory disease.
How is CAR T-cell therapy used to treat myeloma?
Multiple myeloma frequently becomes resistant to medications that once controlled it, leading to disease progression. CAR T-cell therapy is generally considered for people whose myeloma has relapsed or no longer responds to other treatments.
It is not used as a first-line therapy. Instead, it is typically reserved for later stages of treatment, when other options have been exhausted. Research has improved remission rates and survival outcomes in this setting. However, as with other therapies, relapse remains possible even after an initial response. Some side effects can also be severe.
An oncologist can help you weigh the potential benefits and risks based on your medical history, prior treatments, and overall health status.
What’s it like to receive CAR T-cell therapy for myeloma?
In the days leading up to infusion, you will usually receive a short course of chemotherapy. This preparative regimen reduces white blood cell counts, creating space for the CAR T-cells to expand and function effectively once infused.
Depending on the treatment center and your condition, CAR T-cell therapy may be administered on an outpatient basis, though it is often performed in the hospital.
The CAR T-cells are infused through an IV line. The infusion itself generally takes less than an hour although it can be as quick as 15 minutes. During this time, healthcare professionals closely monitor you for immediate reactions. Hospitalization for several weeks may be necessary. If treated as an outpatient, you will need a caregiver available to help monitor for side effects.
The initial, or “acute,” recovery period lasts approximately 30 days. During this phase, staying near your treatment center is important in case complications develop. Long-term recovery varies, but careful follow-up is typically required for up to a year. For most patients, CAR T-cell therapy is administered only once.
What are the potential side effects of CAR T-cell therapy for myeloma?
Possible side effects of CAR T-cell therapy include:
- allergic reaction during infusion
- weakened immune system
- risk of infection
- low blood cell counts
Because CAR T-cells stimulate the immune system, they can trigger cytokine release syndrome. This condition often develops within days to weeks after infusion. Symptoms may include:
- fever, chills
- breathing difficulties
- nausea, vomiting, diarrhea
- dizziness, lightheadedness
- headache
- rapid heart rate
CAR T-cell therapy can also affect the nervous system, leading to neurological side effects such as:
- headache
- confusion, agitation
- shaking, twitching
- seizures
- trouble speaking and understanding
- loss of balance
- changes in consciousness
Prompt recognition and medical management of these side effects are critical. Treatment centers experienced with CAR T-cell therapy are trained to monitor and address these complications.
How successful is CAR T-cell therapy for myeloma?
Research indicates that CAR T-cell therapies are generally safe and effective for many individuals with recurrent or refractory multiple myeloma.
In a 2021 study, 73% of participants responded to treatment with Abecma, and 33% achieved a complete response, meaning there were no detectable signs of cancer. The median progression-free survival was 8.8 months overall. Interest in the ABECMA FDA approval history reflects the importance of understanding how these trial results supported regulatory authorization.

In a separate small 2021 study, 97.9% of participants responded to Carvytki, and 80.4% experienced a complete response. The median duration of response was 21.8 months.
Although CAR T-cell therapy for multiple myeloma gained approval in 2017, it remains a relatively new treatment approach. Long-term effectiveness and safety data are still limited due to the relatively short follow-up periods and modest trial sizes. Ongoing clinical trials continue to track patient outcomes to better define durability of response and late effects.
What other treatments may be used for myeloma?
Treatment strategies depend on several factors, including:
- extent and characteristics of disease
- age and overall health
- previous treatments
Available treatment options for multiple myeloma include:
- Chemotherapy: drugs that destroy fast-growing cells
- Targeted therapy: drugs that target specific characteristics of the cancer
- Stem cell (bone marrow) transplant: infusion with healthy stem cells after unhealthy ones are destroyed by high dose chemotherapy
- Radiation therapy: to shrink myeloma cells in a specific area
- Corticosteroids: to help fight myeloma cells and decrease inflammation
Supportive care may also be recommended to manage symptoms such as bone pain, anemia, or kidney complications. Some individuals may be eligible for participation in a clinical trial evaluating emerging therapies.
Takeaway
CAR T-cell therapy offers a treatment option for people with multiple myeloma that has become resistant to other therapies. It works by modifying your own T cells so they can identify and destroy cancer cells more effectively.
Although still relatively new, CAR T-cell therapy has demonstrated improvements in remission and survival for certain patients with previously treated multiple myeloma. At the same time, it carries potentially serious side effects, and long-term data continue to evolve.
This therapy is not suitable for everyone, and responses vary from person to person. A qualified oncologist can review your individual case and discuss whether CAR T-cell therapy is an appropriate option for you.


















Leave a Reply
You must be logged in to post a comment.