Gene therapy has emerged as a promising scientific approach that may one day offer a transformative treatment for Duchenne muscular dystrophy (DMD). However, early findings from clinical trials have been more modest than researchers initially anticipated. Studies are still underway to better understand its long-term safety, effectiveness, and the overall Cost of gene therapy for families considering this option.

Duchenne muscular dystrophy (DMD) is a genetic disorder characterized by progressive muscle weakness, most commonly affecting children assigned male at birth. Symptoms gradually worsen over time, and at present, there is no established cure.
Because DMD stems from a genetic mutation, therapies designed to correct or compensate for the faulty gene — referred to as gene therapy — may provide a targeted treatment strategy. Several investigational gene therapies for DMD are currently in various stages of development.
This article explains how gene therapy works, reviews its potential role in treating DMD, and explores important considerations such as eligibility, effectiveness, and the Cost of gene therapy.
Can gene therapy help treat Duchenne muscular dystrophy?
Gene therapy is an innovative medical treatment aimed at repairing, replacing, or modifying a defective gene to address the underlying cause of a disease.
DMD is associated with mutations in the dystrophin gene, one of the largest genes in the human genome. These mutations impair the body’s ability to produce dystrophin, a protein essential for maintaining muscle cell integrity. Without sufficient dystrophin, muscle fibers gradually deteriorate and weaken.
In DMD, gene therapy works by repairing or substituting the defective sections of the dystrophin gene so that the body can produce a functional version of the protein. If administered during a critical developmental window, this approach could theoretically slow or halt disease progression.
Despite its potential, most gene therapy treatments for DMD remain in clinical development. Additional research is required to confirm long-term safety outcomes, durability of benefit, and overall therapeutic impact.
Gene therapy drugs for Duchenne muscular dystrophy
In the summer of 2023, the Food and Drug Administration (FDA) granted accelerated approval to delandistrogene moxeparvovec (Elevidys), a single-dose gene therapy developed by Sarepta Therapeutics. Administered using a viral vector, Elevidys enables the body to produce a shortened form of the dystrophin protein.
More recently, the company released results from a 52-week randomized, placebo-controlled phase 3 clinical trial involving 125 male children with DMD. Although Elevidys did not achieve a statistically significant improvement in its primary outcome, it did meet certain secondary endpoints.
Additional gene therapies are also advancing through clinical trials. One example is fordadistrogene movaparvovec, developed by Pfizer and currently being evaluated in a phase 3 clinical trial.
Who is eligible for gene therapy for Duchenne muscular dystrophy?
According to the FDA, Elevidys is approved for male children with DMD who meet specific eligibility requirements:
- are 4–5 years old
- are ambulatory (walking)
- have a confirmed dystrophin gene mutation
- do not have antibodies against the adeno-associated virus (AAV) rh74 vector
- do not have a deletion in exon 8 or exon 9 of the dystrophin gene
Exons are segments of genes responsible for coding proteins. The dystrophin gene contains 79 exons.
Children with DMD may face a heightened risk of serious side effects if they have:
- liver problems
- mutations in exons 1–17 or 59–71 of the dystrophin gene
Careful genetic testing and clinical evaluation are essential before proceeding with gene therapy treatment.
What is the procedure for gene therapy for Duchenne muscular dystrophy?
A healthcare professional will first determine whether your child meets the medical criteria for gene therapy.

Before treatment, your child will undergo blood tests to evaluate liver function, platelet levels, and troponin-I. In the days leading up to the infusion, a corticosteroid regimen is typically prescribed to lower the risk of an immune response.
Elevidys is administered as a single intravenous (IV) infusion over approximately 1–2 hours. Following the procedure, weekly monitoring for side effects is required for at least 3 months. Ongoing follow-up helps clinicians track liver enzymes, immune reactions, and overall health status.
How effective is gene therapy in treating Duchenne muscular dystrophy?
The overall effectiveness of gene therapy for DMD remains under evaluation and may differ between specific treatments.
Data published by Sarepta Therapeutics from the phase 3 clinical trial of Elevidys indicated that the therapy did not meet its primary efficacy endpoint. However, compared with placebo, participants receiving Elevidys demonstrated:
- a modest but not statistically significant improvement in movement
- improved time to rise from the floor
- better performance in timed 10-meter walking tests
Long-term follow-up studies are needed to determine whether these functional gains persist and translate into meaningful clinical benefits over time.
How does gene therapy compare to other treatments for Duchenne muscular dystrophy?
Corticosteroids such as prednisone remain the standard of care for DMD. These medications reduce inflammation and can help preserve muscle strength for a longer period than would occur without treatment.
However, corticosteroids must be taken consistently, and prolonged use may lead to significant side effects. Importantly, they are supportive therapies, meaning they help manage symptoms but do not address the genetic cause of DMD.
By contrast, gene therapy is designed as a one-time intervention that directly targets the underlying dystrophin gene mutation. While this targeted approach is scientifically compelling, uncertainties remain regarding durability, safety, and the Cost of gene therapy compared with long-term standard treatment. Broader discussions about Gene therapy pricing models highlight how payment structures and insurance coverage may influence patient access.
Recently-approved targeted therapies for DMD
The FDA has also approved several exon-skipping therapies for DMD. These medications focus on specific genetic mutations and include:
- eteplirsen (Exondys 51)
- golodirsen (Vyondys 53)
- casimersen (Amondys 45)
- viltolarsen (Viltepso)
These therapies are mutation-specific and may only benefit patients with particular exon deletions.
How much does gene therapy for Duchenne muscular dystrophy cost?
The Cost of gene therapy for DMD can reach several million dollars for a single treatment. Elevidys is priced at $3.2 million per dose. Because it has FDA approval, insurance providers may offer coverage, though out-of-pocket expenses and authorization requirements can vary. Families often research resources such as What is the cheapest gene therapy to better understand affordability and financial planning.
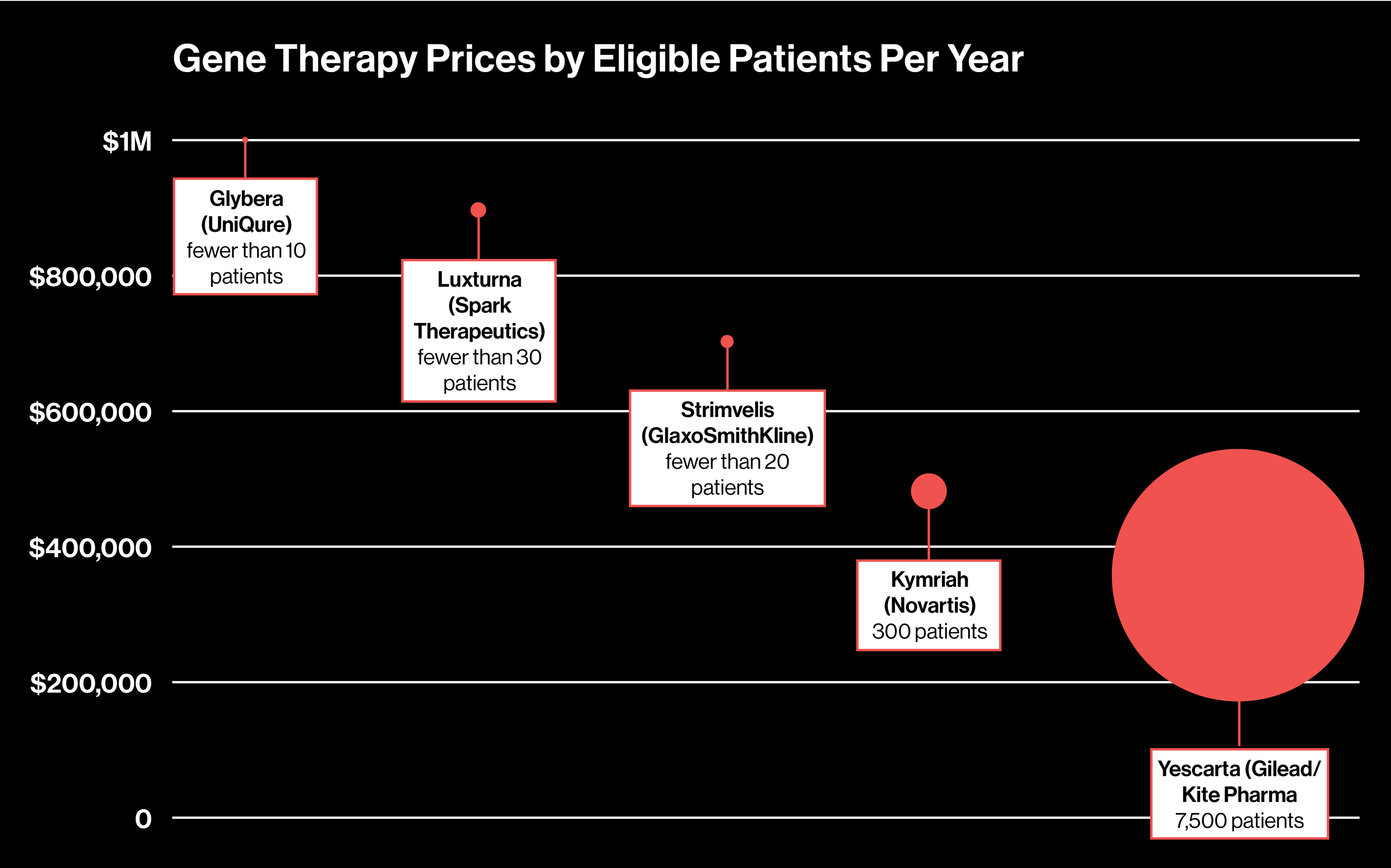
For comparison, a 2022 study estimated that the cumulative direct cost of conventional DMD treatments could reach up to $2.3 million over 20 years. While gene therapy involves a high upfront expense, traditional therapies may accumulate substantial long-term costs related to medications, monitoring, and supportive care.
Beyond financial considerations, families must also weigh potential clinical benefits, treatment durability, and quality-of-life outcomes when evaluating therapeutic options.
Frequently asked questions
Below are answers to some common questions about gene therapy for DMD.
Can gene therapy cure Duchenne muscular dystrophy?
Gene therapy has been described as a potential cure for DMD. Although it shows Great promise, important questions remain about its long-term safety, durability, and real-world effectiveness.
Is gene therapy FDA approved for Duchenne muscular dystrophy?
The FDA granted accelerated approval to Elevidys in 2023, making it the first gene therapy for DMD. The Accelerated approval pathway is designed to expedite access to treatments for life threatening medical conditions.
Can gene therapy treat Becker muscular dystrophy or limb-girdle muscular dystrophy?
Research into gene therapies for other forms of muscular dystrophy, including Becker muscular dystrophy and limb-girdle muscular dystrophy, is ongoing. At this time, evidence is insufficient to confirm their effectiveness.
Takeaway
Gene therapy for DMD represents a rapidly evolving treatment approach aimed at correcting dystrophin gene mutations.
Delandistrogene moxeparvovec (Elevidys) received accelerated approval from the FDA in 2023. However, recent phase 3 trial results indicate that its effectiveness may be more limited than initially expected.
Additional gene therapies are in clinical development. If your child has DMD, discussing available treatments — including potential benefits, risks, and the Cost of gene therapy — with your doctor can help guide informed decision-making.




















Leave a Reply
You must be logged in to post a comment.